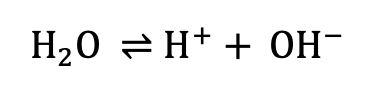In manufacturing environments, various cleaning agents are used to remove contaminants such as rust, grease, rust preventive oils, and cutting fluids. Cleaning agents can be broadly classified into two main types: solvent-based and water-based. Water-based cleaners are further classified into three types by pH: acidic, neutral, and alkaline. Each type has different characteristics and applications.
Basic Principles of Acidity and Alkalinity
First, let us clarify what gives rise to the properties known as acidity and alkalinity. The acidity or alkalinity of an aqueous solution is determined by the balance between hydrogen ions (H⁺) and hydroxide ions (OH⁻). Hydrogen ions (H⁺) are responsible for acidic properties, while hydroxide ions (OH⁻) are responsible for alkaline properties.
Water is chemically represented as H₂O. However, upon closer examination of water molecules, we observe that they undergo the following reaction.
Unlike typical chemical reactions, the arrow points in both directions. This indicates that the reaction proceeds both from left to right and from right to left. In water, water molecules do not always exist solely as H₂O; they continuously dissociate into H⁺ and OH⁻ ions, while these ions recombine to form H₂O again.
Therefore, if we observe water at a microscopic level at any given moment, we will find not only H₂O molecules, but also small amounts of H⁺ and OH⁻ ions present.
Now, in the equation shown above, H⁺ and OH⁻ are produced in equal amounts. In this state, the ions responsible for acidity and the ions responsible for alkalinity are present in equal concentrations. Therefore, the solution is neither acidic nor alkaline—it is neutral.
Conversely, an acidic solution has a higher concentration of H⁺ ions, while an alkaline solution has a higher concentration of OH⁻ ions.
Ion Balance
- Acidic: High concentration of H⁺ ions
- Neutral: Equal concentrations of H⁺ and OH⁻ ions
- Alkaline (Basic): High concentration of OH⁻ ions
What is pH?
pH is a commonly used indicator to express the strength of acidity or alkalinity. The term pH stands for “potential of hydrogen,” and it represents the concentration of hydrogen ions (H⁺), which are the source of acidity in an aqueous solution.
Without going into detail, the concentration of H⁺ ions in a neutral aqueous solution corresponds to a pH of 7. Using pH 7 as the reference point, the value becomes lower as the solution becomes more acidic, and higher as the solution becomes more alkaline.
pH Scale Examples

Because pH is a logarithmic expression of the concentration of hydrogen ions (H⁺), highly concentrated acidic solutions can have a pH of 0 or even negative values. However, such extremely strong acids are rarely encountered in everyday life.

How does the pH of a cleaning solution affect its cleaning performance?
Characteristics and Applications of Acidic Cleaners
Acidic cleaners are most effective when removing the following types of contaminants:
- Rust formed on metal surfaces
- Scale deposits accumulated inside pipes
- Concrete and mortar residues
- Urinary scale buildup in toilets

However, acidic cleaners also have certain disadvantages, so it is important to understand their limitations and use them appropriately.
Disadvantages of Acidic Cleaners
One disadvantage of acidic cleaners is that they can promote metal corrosion. While acids are effective at removing rust, any residual acid left on the surface after cleaning can accelerate the formation of new rust. Therefore, it is essential to thoroughly rinse off the acid after rust removal. In addition, once rust is removed, the bare metal becomes more susceptible to corrosion, making protective measures such as coatings or paint necessary.
Extra caution is also required when using acids to remove scale buildup inside pipes. To clean pipes effectively, the cleaning solution must be circulated while maintaining a sufficiently low pH. As a result, a large volume of low-pH wastewater is generated after the cleaning process.
However, wastewater treatment can be extremely challenging. Diluting acidic wastewater with water to reduce its acidity requires a very large volume of water. For example, to dilute wastewater with a pH of 1 to near neutral conditions (pH 6), it would need to be diluted approximately 100,000 times. Because such dilution is impractical, neutralization with specialized chemicals is necessary. Pipe cleaning with acids is typically a much larger and more complex task than routine maintenance work. Therefore, it is essential to conduct thorough advance planning to minimize environmental impact and reduce wastewater volume.
When to Use Alkaline Cleaners
Alkaline cleaners are most effective when removing the following types of contaminants:
- Oil and grease deposits
- Fat and oil buildup in kitchens and food processing facilities
- Protein-based blockages, such as hair clogging pipes
- Dirt and contaminants attached to aluminum fins in heat exchangers.

Among these, oil, grease, and fat can also be removed using neutral cleaners or solvent-based cleaners. However, when it is necessary to dissolve protein-based contaminants, an alkaline cleaner is the most appropriate choice.
Disadvantages of Alkaline Cleaners
Unlike acidic cleaners, which can easily cause corrosion, alkaline cleaners are less likely to promote rust. However, alkaline cleaners also have their own disadvantages. These are closely related to the effect alkaline cleaners have on proteins, as mentioned earlier.
One of the key characteristics of alkaline substances is their ability to dissolve proteins. A familiar example is household bleach—if you accidentally touch it, the skin may feel slippery. This occurs because the alkaline substance dissolves the proteins on the skin’s surface. As a result, using alkaline cleaners without proper protective equipment can lead to skin irritation and, in severe cases, even eye damage or blindness.
Of course, this risk is not limited to alkaline cleaners; many chemical products require proper handling. However, compared to acids, alkaline substances are often perceived as less dangerous because they tend to have less noticeable odors. This can lead to underestimating the necessary safety precautions.
In addition, as with acidic cleaners, wastewater treatment must also be considered when alkaline cleaners are used in large quantities. Aluminum dissolves in alkaline solutions, which is why alkaline cleaners are used to clean aluminum fins in air conditioning systems. Since this process generates a large volume of wastewater, careful planning and preparation are required, similar to acid cleaning of piping systems.
Summary
In this article, we focused on acidity and alkalinity to explain the characteristics of different types of cleaning agents. Both acidic and alkaline cleaners have advantages and disadvantages, and it is important to understand their properties when selecting and using them appropriately.
NCH is committed to providing the most effective solutions to address our customers’ maintenance challenges.
 Australia & NZ
Australia & NZ 中国
中国 India
India Indonesia
Indonesia 日本
日本 한국
한국 Malaysia
Malaysia Philippines
Philippines Singapore
Singapore 台灣
台灣 ประเทศไทย
ประเทศไทย Tiếng Việt
Tiếng Việt